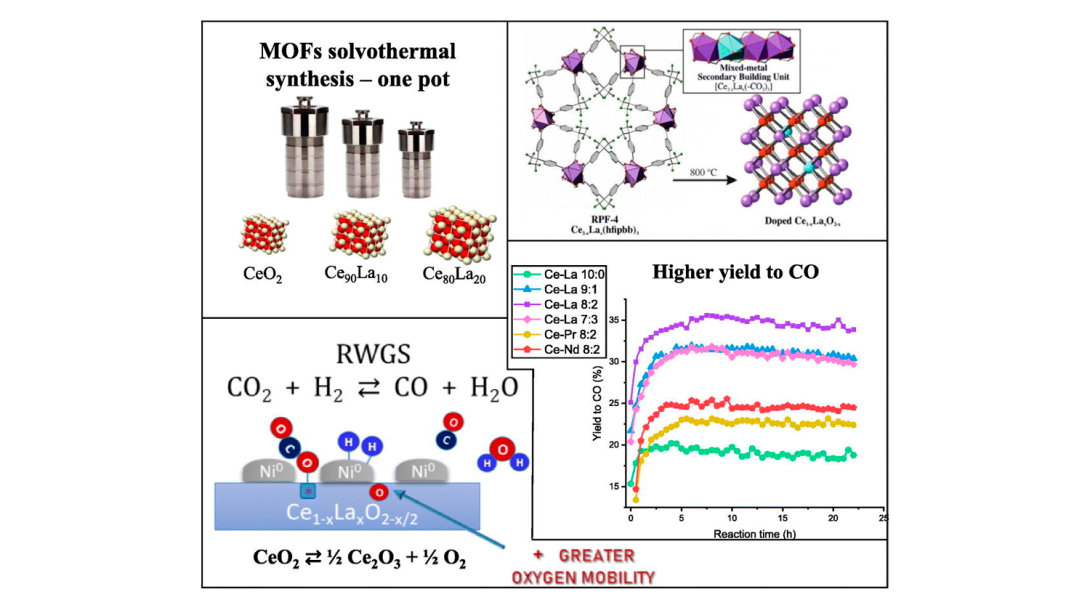
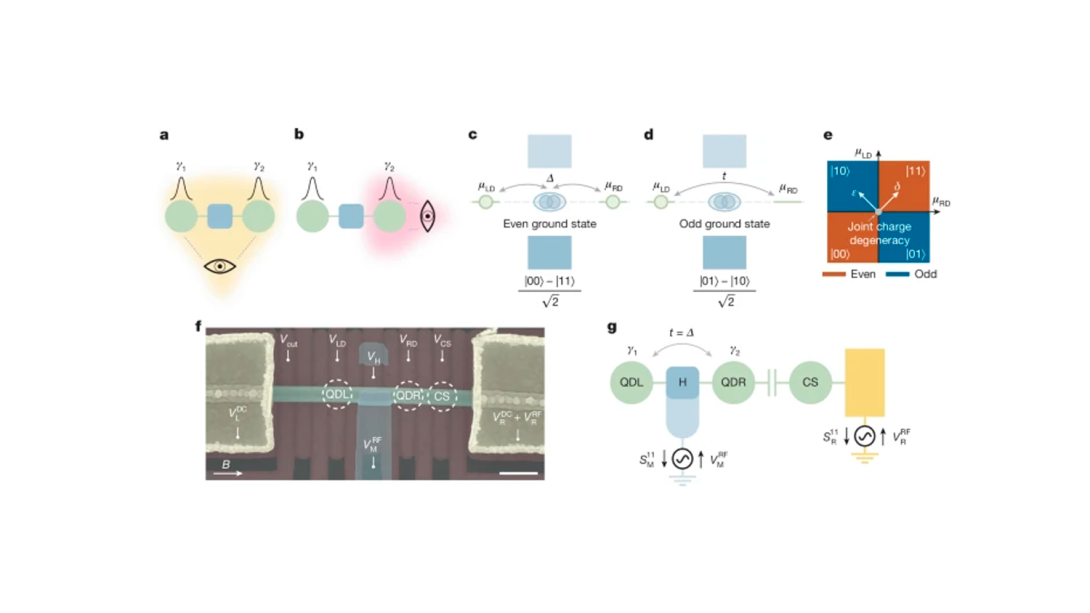
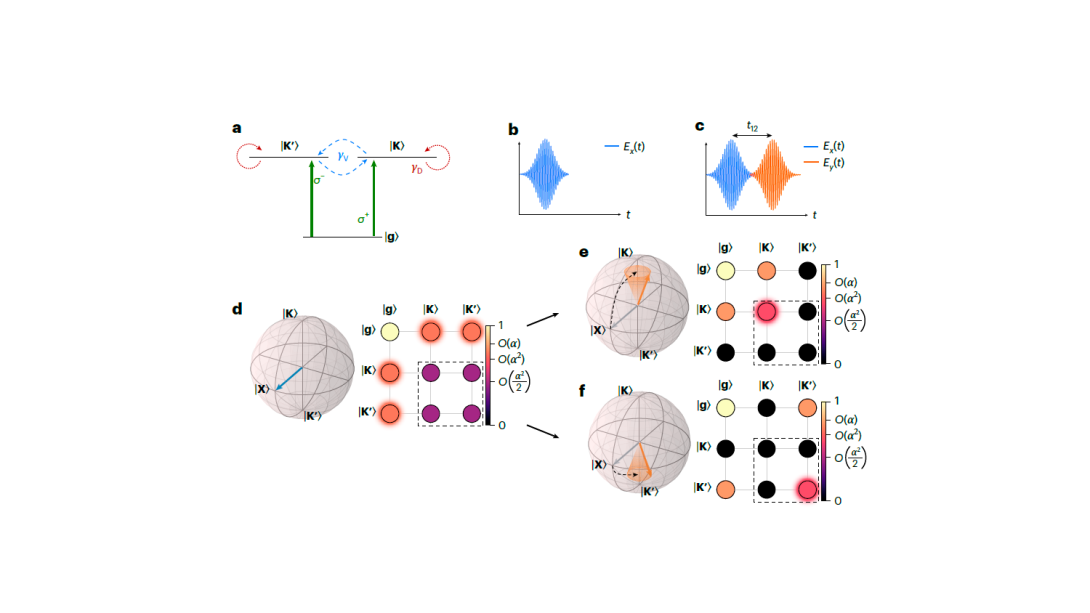
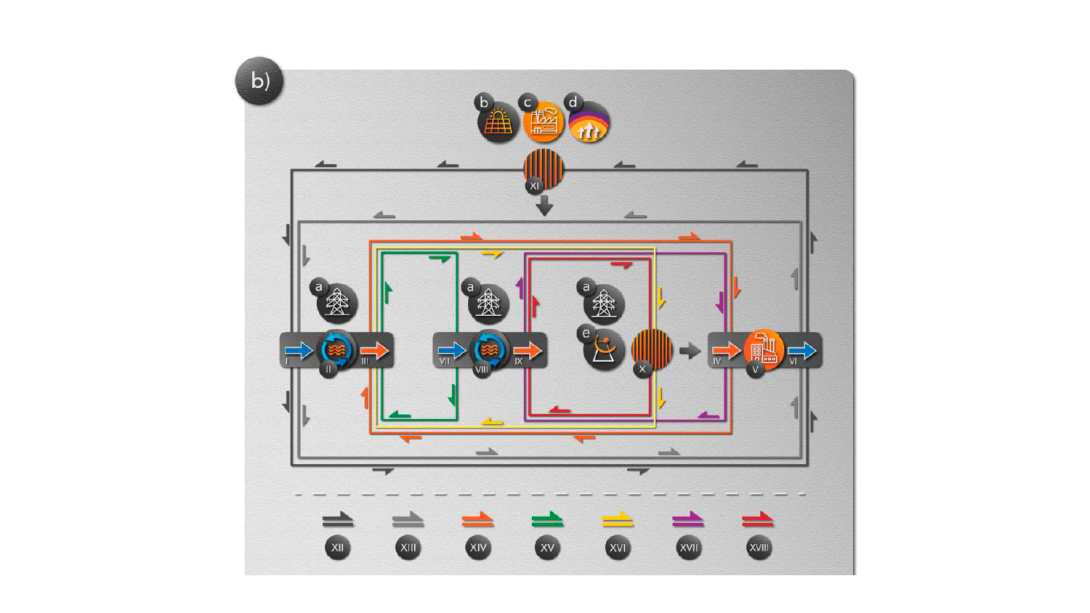
Wafer-scale films of two-dimensional materials via roll-to-roll mechanical exfoliation
Yigit Sozen, Thomas Pucher, Bhagyanath Paliyottil Kesavan, Nuria Jiménez-Arévalo, Julia Hernandez-Ruiz , Zdenek Sofer, Carmen Munuera, Juan J. Riquelme, Andres Castellanos-Gomez*