Researchers create a new cancer strategy that achieves greater cell death with less chemotherapy thanks to nanomedicine
A study led by the Madrid Institute of Materials Science (ICMM-CSIC) demonstrates the potential of cancer therapies that, by combining two types of hyperthermia (heat treatments) with chemotherapy using nanoparticles, allow for reduced drug doses. The study, published and selected as the cover story in the journal Advanced NanoBiomed Research, advances toward more effective and accessible treatments for tumors.
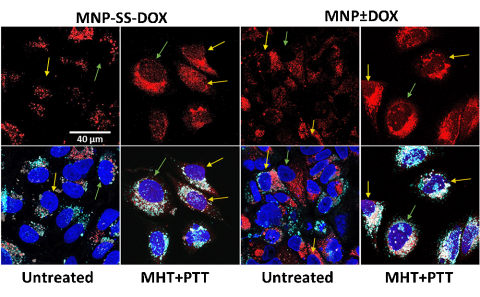
The research team has created magnetic nanoparticles (one millionth of a centimeter in size) to which they have added a chemotherapeutic drug, doxorubicin. To carry out their new treatment, called trimodal – because it combines three synergistic approaches against cancer – they applied magnetic stimulation to the nanoparticles, which generates heat (magnetic hyperthermia), while simultaneously applying a near-infrared laser, which also produces heat (photothermal treatment). Additionally, "we have made the nanoparticles release the drug precisely when they receive this heat, which is known as localized chemotherapy under clinically safe conditions," explains Ana Espinosa, a CSIC researcher at the Institute of Materials Science of Madrid (ICMM-CSIC) and the study's lead author.
"By activating pH-sensitive drug release and synergistic heating inside cancer cells, these nanoparticles achieve potent destruction of tumor cells while minimizing systemic toxicity," the scientist continues.
Espinosa notes that cancer cells are particularly sensitive to heat, so with this treatment, they aim to create a 'heat trap' that destroys those tumor cells. "We seek to produce a therapeutic effect while allowing us to reduce toxic doses to healthy tissues," the researcher states.
Since using each technique separately does not safely achieve the required temperature, this scientist and her team sought a synergistic effect: "You add up the effects, and this also allows you to reduce the intensity of the application parameters. We can lower the laser intensity and also the magnetic field, making the treatment less aggressive," she explains.
The study, which primarily focused on demonstrating the potential of the trimodal treatment, worked with iron oxide magnetic nanoparticles because this substance is well absorbed by the body, degrades naturally, and at these doses is non-toxic.
"We have achieved cell death that is up to 70% more efficient than with individual therapies," Espinosa details. The studies, so far, have been conducted on in vitro models of breast cancer tumor cells, although they could be extended to any type of cancer and are replicable on a larger scale.
The findings "highlight the potential clinical application of multifunctional nanoparticle systems for specific and low-toxicity cancer therapies, advancing toward more effective and accessible treatments," the researcher concludes.
The work is a collaboration between the Institute of Materials Science of Madrid (ICMM-CSIC), IMDEA Nanociencia Institute, the Curie Institute (France), and the Institute of Ceramics and Glass (ICV-CSIC).
Reference:
Rosalía López-Méndez, Nuria Lafuente-Gómez, Eva Céspedes, Mónica Dhanjani, Marina París-Ogáyar, Francisco José Terán, Aida Serrano, Julio Camarero, Gorka Salas, Claire Wilhelm, Álvaro Somoza, Ana Espinosa*. Tri-Modal Anticancer Strategies with Doxorubicin-Loaded Iron Oxide Nanoparticles Integrating Chemo and Magneto-Photothermal Therapeutic Effects. Advanced NanoBiomed Research. DOI: https://doi.org/10.1002/anbr.70084
Instituto de Ciencia de Materiales de Madrid (ICMM)
Sor Juana Ines de la Cruz, 3
Cantoblanco, 28049
Madrid, España
Telephone: (+34) 91 334 90 00
Email: @email
Communication Office: @email

Acknowledge the Severo Ochoa Centres of Excellence program through Grant CEX2024-001445-S/ financiado por MICIU/AEI / 10.13039/501100011033

Contacto | Accesibilidad | Aviso legal | Política de Cookies | Protección de datos
