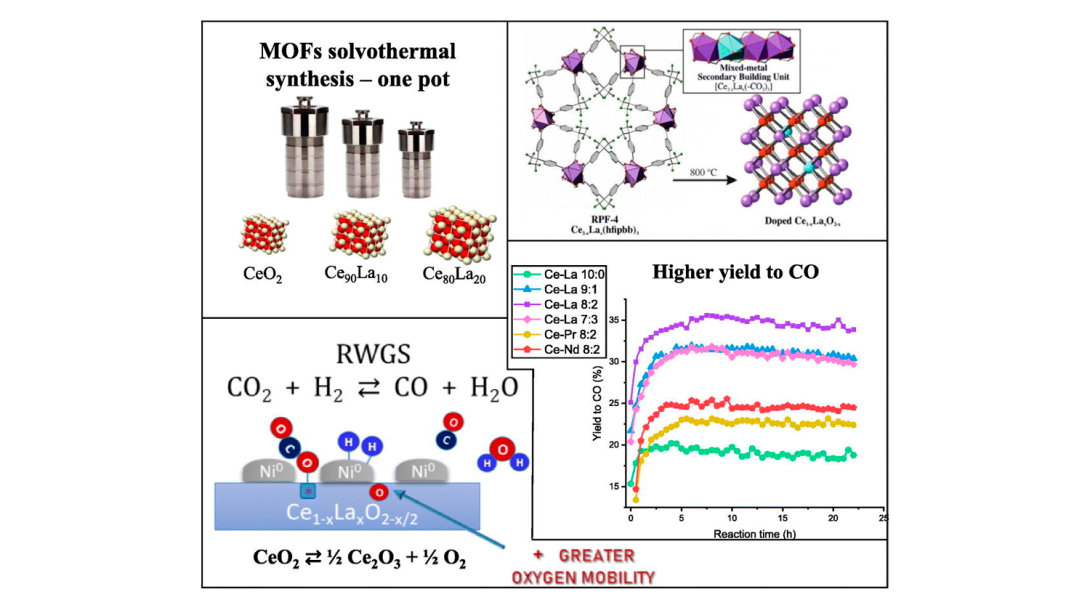
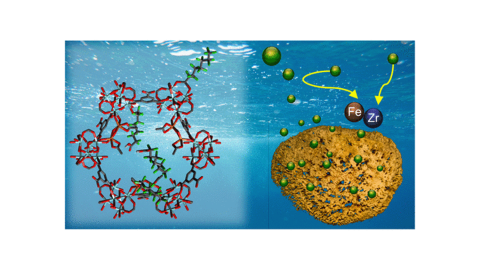
Atomically Precise Engineering of Synergistic Binding Sites in a Zirconium Metal–Organic Framework for the Capture of Perfluorooctanoic Acid
Sergio Marugán-Benito, Michalis Vlachos, Lutz Ahrens, Miguel Roselló-González, Carlo Marini, Jordi Prat Albert, Andreas Mavrandonakis*, Edward Loukopoulos*, Ana E. Platero-Prats*
Journal of the American Chemical Society
10000
News
Welcome
5000
Agenda
Seminars at ICMM
Instituto de Ciencia de Materiales de Madrid (ICMM)
Sor Juana Ines de la Cruz, 3
Cantoblanco, 28049
Madrid, España
Telephone: (+34) 91 334 90 00
Email: @email
Communication Office: @email

Acknowledge the Severo Ochoa Centres of Excellence program through Grant CEX2024-001445-S/ financiado por MICIU/AEI / 10.13039/501100011033

Contacto | Accesibilidad | Aviso legal | Política de Cookies | Protección de datos