UV-Induced Mechanism of Aliphatic Cleavage in the Interstellar Medium
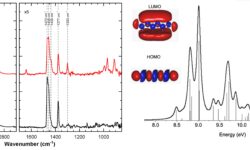
In the interstellar medium (ISM), UV processing leads to significant chemical transformations in hydrocarbons. Aliphatic hydrocarbons such as alkanes, commonly formed in evolved stars, undergo dehydrogenation primarily through UV-induced photocleavage of C–C bonds rather than direct hydrogen elimination. This study combines experimental data and ab initio calculations to understand how…
Front cover in ACS Applied Energy Materials
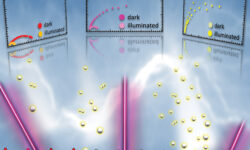
One of the primary challenges associated with the use of titanium dioxide as a photocatalyst lies in its wide bandgap. In our research, we addressed this issue by employing a thermal treatment in atomic hydrogen plasma to reduce TiO2. This process led to an enhancement in the material’s light absorption…
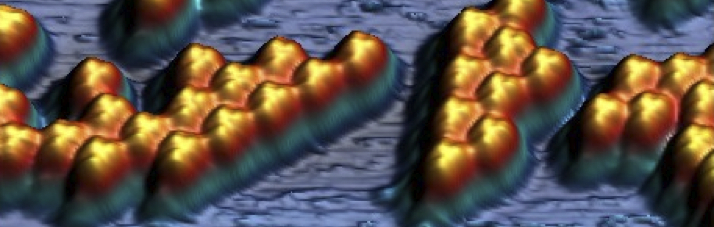
Unraveling the mechanisms of Thermal Dehydrogenation of n-Octane on Pt(111) at the atomic scale
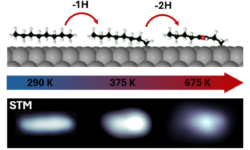
In our article published recently in Nanoscale, we focus on the thermal dehydrogenation of n-octane (n-C8H18) on the catalytic Pt(111) surface in ultra-high vacuum. Our results shed light on the mechanisms behind the initial stages of this industrially relevant reaction, paving the way for the design of efficient dehydrogenation catalysts.…
Development of a Graphene SG-FET aptasensor able to detect attomolar concentrations of hepatitis C virus core protein
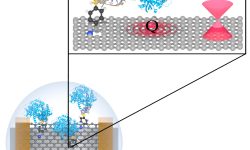
Biosensors based on graphene field-effect transistors have become a promising tool for detecting a broad range of analytes. However, they lack the stability and reproducibility required to step into biotechnological and biomedical applications. ESISNA group has used a promising physical graphene functionalization protocol which allows atomically-precise highly-controlled covalent functionalization, preserving…
The influence of reaction conditions on the growth of MOFs on surfaces
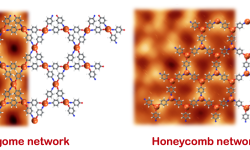
The importance of the reaction conditions has been confirmed in this study, where following an on-surface synthesis approach on the same surface and with the same precursor, two different metal-organic frameworks were obtained. The distinction comes from the different synthesis protocol of the controlled growth of both metal-organic nanostructures. Thus,…
Front cover in Chemistry – A European Journal!

Host-guest interactions are of paramount importance in supramolecular chemistry and in a wide range of applications. Particularly well known is the ability of cucurbit[n]urils (CB[n]) to selectively host small molecules. We show that the charge transfer and complexation capabilities of CB[n] are retained on the surface of 2D transition metal…
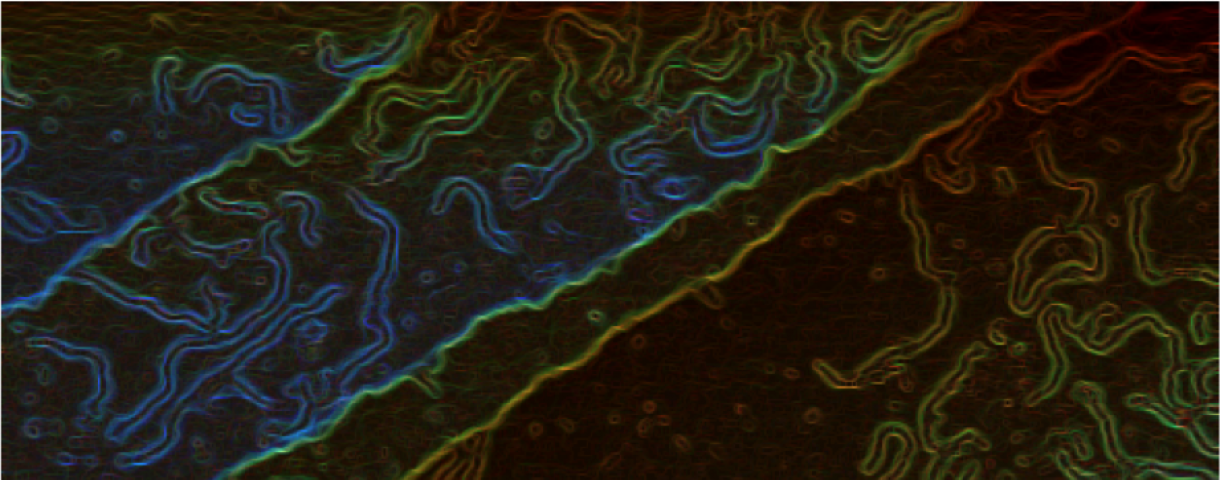
Why individual heptagonal rings are not stable on surfaces?
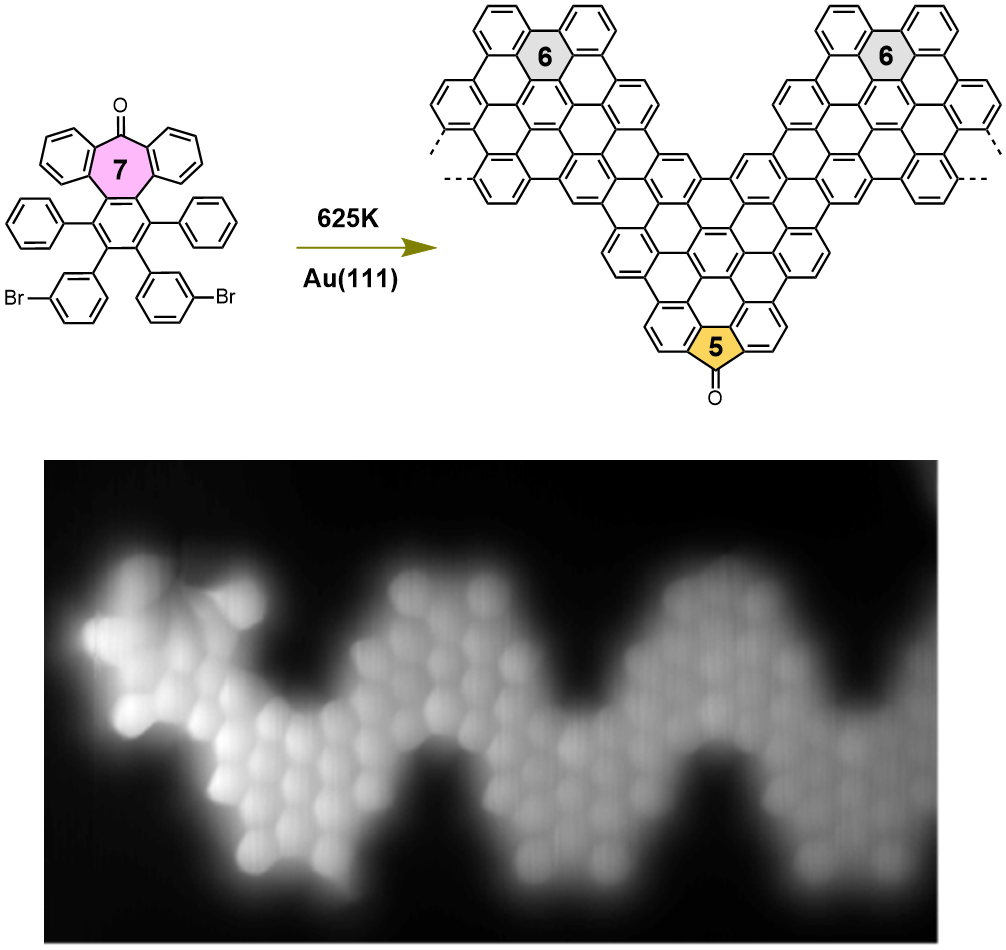
Graphene nanoribbons, GNRs, – narrow stripes of graphene – are predicted to exhibit attractive electronic properties for the fabrication of nanoscale electronic devices. This 1D material shows semiconducting properties that can be tuned by modifying its length, width, or topology border; but also, with the presence of defects in form…
First European users of Stardust at ICMM!
PhotoSurf: ESISNA starts new research project side by side with FRONCAT

ESISNA, research group in Material Science, and FRONCAT, research group in Chemistry, will work together on PhotoSurf, a synergy I+D project of the Comunidad de Madrid. Both groups will join forces to mimic the photosynthesis process of plants and generate new materials using light. Project FOTOSURF-CM, ref. Y2020/NMT-6469